
Apple Flavour Compounds
Nigel Deacon
There is no layman's guide to apple flavours and the compounds which cause them. This is my attempt to compile one. It is not authoritative; I am merely an apple enthusiast with a knowledge of Chemistry.
There are many ways of classifying apple flavours. The main divisions are SWEET, SOUR and BITTER. To a first approximation, SWEET and fruity flavours come from sugars and esters, those which are SOUR come from acids, and BITTER flavours come from a range of compounds known as tannins or polyphenols.
Esters and sugars are probably the main contributors to apple flavour, modified by the presence of carboxylic acids, alcohols and tannins. Often other compounds are present too, which can make a variety easy to recognize if it has a distinctive taste.
Part of an apple's flavour comes from its scent and the volatile organics released on consuming it: esters (fruity), alcohols (fruity or earthy), aldehydes (appley and slightly grassy & bitter), and ethers and terpenes (scented oils).
There are flavours which stand out in only a few varieties: honey, fennel, aniseed, strawberry, raspberry, melon, blackcurrant, banana, parma violets, and certain bitter tastes. Bitterness is often strongest in or just below the skin; the bitter compounds dissolve easily in the waxy skin layer.
A lot of research has been done on the biochemistry of apple storage, and the effect it has on apple flavours. Most of the published papers are impenetrable to those without specialist knowledge of the subject area. The paper 'Factors affecting apple aroma / flavour volatile concentration' by by Dixon and Hewitt, 2000 is useful if you know enough science to follow it. (ref 1)
The overall flavour of an apple is dependent on the balance of bitterness, sweetness and acidity, and the presence or absence of volatile aromatic compounds. Early apples have few aromatics and little depth of flavour, but they often have more acidity and sweetness. (e.g. Discovery, Merton Knave, Almata)
Cooking apples have large amounts of malic acid which assists the apple's breakdown during cooking but which masks other flavours; this is why most cookers are unpalateable as dessert apples. On storage or on cooking, the malic acid content decreases. Some varieties of apple too acidic to eat straight away can be eaten raw if they can be stored. Even Bramleys can be used as a dessert apple in some years for a period of about three weeks, after the acidity has dropped and before the skins go tough.
Late dessert apples have lower water content and higher proportions of sugars, esters and aromatics; the apples with the richest flavours are almost all mid to late-ripening varieties. (Ribston, Wickson, Cox, Rubinett, Allen's Everlasting, etc)
All apples contain a selection of the compounds below, but the proportions in which they appear are infinitely variable. This is why no two varieties taste quite the same.
So: the different flavours:
SWEET
The sugars in ripe apples are mainly sucrose, glucose and fructose in the approximate ratio 1:1:2. Apples tasting of honey often have more than the usual amount of fructose. This is seen in Golden Delicious on a good day, and the 'honey apple' found recently in Sutton in the Elms, Leics. Certain esters (and a lack of acid) also accentuate the honey flavour. The structures are too well-known to merit inclusion here.
SOUR
All acids release hydrogen ions, H+, and all hydrogen ions taste the same - sour. Some of the undissociated acid molecules have characteristic flavours too; we all have experience of vinegar, which contains acetic acid (not present in apples at a concentration we can taste) but the acids in apples are similar; they tend to be carboxylic acids with an even number of carbon atoms and characteristic tastes.
At the concentrations found in apples, they have no aroma. Malic acid is the main acid present in apples. The more sour the apple, the more malic acid it contains. Bramleys contain about 0.9% malic acid by weight; some cider apples contain about twice that amount. Acids with little or no tartness contain hardly any malic acid at all.
Other acids are found in apples, but to a much smaller degree. There are the other acids of the Krebs (Citric Acid) Cycle familiar to students of Biology: fumaric, pyruvic, oxaloacetic, ketoglutaric, and citric acid itself. Sometimes quinic acid is present, which has a bitter taste and which is involved in the synthesis of aromatic compounds including polyphenols (tannins). It is interesting that tartaric acid (same as malic acid except for an extra hydroxy group) is absent because it is formed using a different metabolic pathway.
Appreciable vitamin C (ascorbic acid) is present in raw apples of certain varieties and contributes a little to the flavour; it also increases the time taken for a cut apple to go brown.
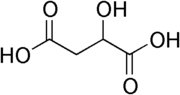 ...
...
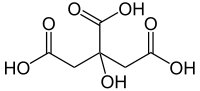
The first structure above is malic acid. The second is citric acid, present as a consequence of the citric acid cycle, which plays an important part in the metabolism of all plants.
FRUITY: ESTERS
A lot of the fruity background taste of apples comes from esters. These are synthesised just under the skin of an apple from the carboxylic acids and alcohols present, under the influence of an enzyme. Of the many esters found in apples: ethyl butanoate, ethyl-2-methylbutanoate, ethyl hexanoate are typical in that they are 'fruity' without imparting a specific flavour character.
Pentyl acetate is in apples and is reminiscent of banana. It is the classic ester preparation done by generations of A-level chemistry students since it smells of pear drops.
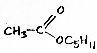
Hexyl acetate (shown below) is pear-like; very similar to pentyl acetate. It contributes part of the aroma of Cox.

Ethyl 2-methy butanoate is reminiscent of strawberries.
The 'red apple' aroma associated with Gala apples is largely hexyl acetate.

The aroma of Worcester Pearmain is largely propyl 2-methyl-butanoate.
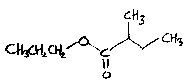
Butyl acetate is pineapple-like. Pineapple flavour occurs in many apples, notably Claygate Pearmain in a good year, and Allington Pippin.
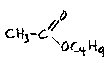
It is interesting that many of these esters when prepared in the laboratory do not smell fruity. Above a certain concentration, they tend to smell rubbery and solvent-like, and a number of them are in fact used as solvents; for example, as paint thinners. It is only when a spot of ester is dissolved in a large excess of water that the fruitiness becomes apparent.
ALCOHOLS
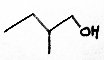
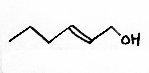
Some apple sensory 'notes' also come from alcohols; principally butan-1-ol (sweet aroma), hexan-1-ol (earthy, unpleasant) and trans 2-hexenol (harmonious, fruity). Propan-1-ol and 2-methyl butan-1-ol are also present, as might have been inferred from the existence of the propyl and 2-methylbutyl esters above.
The structures below are 2-methyl butan-2-ol, hexan-1-ol and trans 2-hexenol.
 ...
...
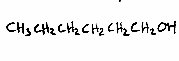 ...
...
ALDEHYDES
The three main aldehydes in apples are ethanal, trans 2-hexenal and hexanal. The sensory impression of ethanal is green / sharp; trans-2-hexenal is green / sharp; harmonious and fruity; hexanal is green / sharp; earthy and grass-like. These are similar to the corresponding alcohols above except that the primary alcohol group CH2OH is replaced by -CH=O
FENNEL & ANISE
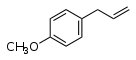
This flavour arises from the presence of an anisole derivative,1-methoxy-4-propenylbenzene, also known as estragole. Chemists will recognize the structure as an ether.It is present in Ellison's Orange, Laxton's Fortune, Fenouillet Gris (hence the name) and others, and imparts an aniseed flavour.
PARMA VIOLETS
A small number of apples contain beta-ionone and its derivatives, based on a terpene-like structure, which give a strange, attractive after-taste of parma violets. In tiny amounts this is pleasant; it is shown to best advantage in West Virginia Sweet (see ref 2) but is present also in Maloni Lilly and to a lesser degree in Scrumptious and the Durrant apple. In large amounts the flavour becomes overpowering and confers a somewhat astringent flavour more suited to a cider apple.

NUTTY FLAVOURS
These are normally associated with Russets, or a well-matured Blenheim Orange. I have not, so far, been able to identify which class of compound is responsible for nutty flavours.
BITTER FLAVOURS
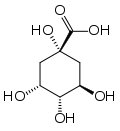
These are usually caused by tannins, or polyphenols. These are synthesized from quinic acid which is shown below. Tannins are complex molecules (there is no single 'tannin' molecule) containing benzene rings, to which are attached several hydroxy groups.
Apples are usually bitter when unripe, and scientists have suggested that this decreases the chance of the fruit being eaten before the seeds are ready. Unripe apples also contain unpalateable amounts of malic acid which would have the same effect.
REFERENCES
1. Dixon, J and Hewitt, E., Factors affecting apple aroma / flavour volatile concentration; New Zealand Journal of Crops and Horticultural Science, 2000, vol 28, pp 155-173
2. Not commercially available, but grown by Leicestershire Heritage Apples. See West Virginia Sweet
ND / Diversity website